- KYPROLIS® (carfilzomib) is indicated in combination with dexamethasone, or with lenalidomide plus dexamethasone, or with daratumumab plus dexamethasone, ... Read More Close
KYPROLIS® demonstrated up to 42 months of mPFS when paired with an anti-CD38 mAb + dex1,2
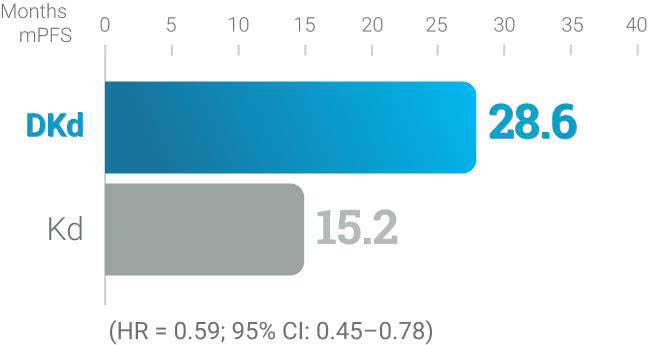
DKd
KYPROLIS® + daratumumab + dexamethasone
CANDOR trial (DKd vs Kd):1,* Randomized, open-
label, multicenter trial of 466 patients who were
randomized 2:1 to receive DKd (n = 312) or Kd
(n = 154) with KYPROLIS® 56 mg/m2 twice weekly.
At a median follow-up of nearly 28 months, mPFS
was 28.6 months for DKd vs 15.2 months for Kd
(HR = 0.59; 95% CI: 0.45–0.78).
Isa-Kd
KYPROLIS® + isatuximab-irfc + dexamethasone
IKEMA trial (Isa-Kd vs Kd):2,3,†,‡ Randomized,
open-label, multicenter trial of 302 patients who
were randomized 3:2 to receive Isa-Kd (n = 179) or
Kd (n = 123) with KYPROLIS® 56 mg/m2 twice
weekly. At a median follow-up of 44 months,
mPFS was 41.7 months for Isa-Kd vs 20.8
months for Kd (HR = 0.59; 95.4% CI: 0.42–0.83).
*As of the primary analysis, with a median follow-up of ~ 17 months, the primary endpoint of improved median PFS was met. Median PFS was not reached for DKd vs 15.8 months for Kd (HR = 0.63; 95% CI: 0.46–0.85; P = 0.0014, one-sided).1,4
†As of the primary analysis, with a median follow-up of 20.7 months, the primary endpoint of improved median PFS was met. Median PFS was not reached for Isa-Kd vs 20.27 months for Kd (HR = 0.55; 95% CI: 0.37–0.82; P = 0.0032).2,3
‡Analysis censoring PFS events occurring > 8 weeks from the last valid disease assessment.2
CI, confidence interval; dex, dexamethasone; HR, hazard ratio; Kd, carfilzomib + dexamethasone; mAb, monoclonal antibody; mPFS, median progression-free survival; PFS, progression-free survival.
DKd KYPROLIS® + daratumumab + dexamethasone
CANDOR (DKd vs Kd):1,†
Randomized, open-label,
multicenter trial of 466
patients who were randomized
2:1 to receive DKd (n = 312) or
Kd (n = 154) with KYPROLIS®
56 mg/m2 twice weekly.
Isa-Kd KYPROLIS® + isatuximab-irfc + dexamethasone
IKEMA (Isa-Kd vs Kd):2,3,‡,§
Randomized, open-label,
multicenter trial of 302 patients
who were randomized 3:2 to
receive Isa-Kd (n = 179) or
Kd (n = 123) with KYPROLIS®
56 mg/m2 twice weekly.
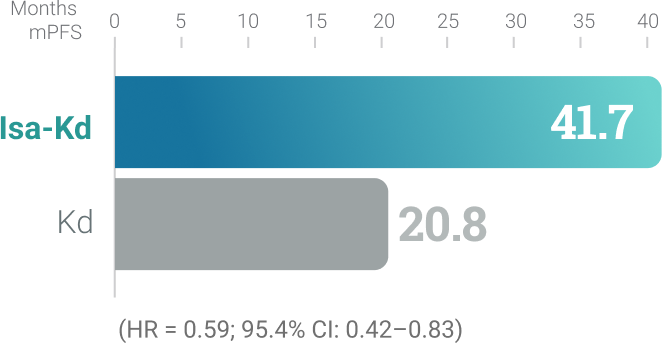
KRd KYPROLIS® + lenalidomide + dexamethasone
ASPIRE (KRd vs Rd):5,**
Randomized, open-label,
multicenter trial of 792 patients
who were randomized 1:1
to receive KRd (n = 396) or
Rd (n = 396) KYPROLIS®
27 mg/m2 twice weekly.§
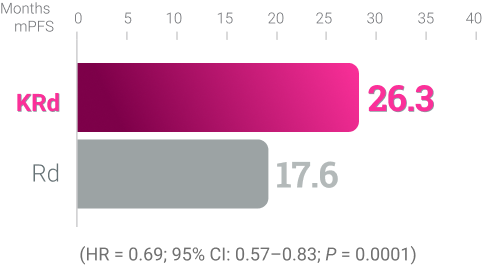
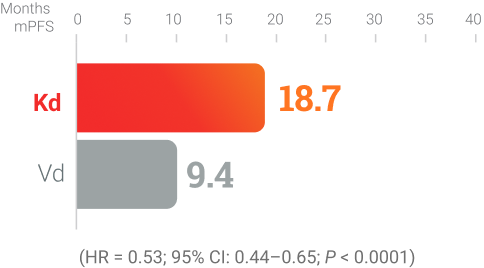
Kd KYPROLIS® + dexamethasone
ENDEAVOR (Kd vs Vd):6
Randomized, open-label,
multicenter trial of 929 patients
who were randomized 1:1
to receive Kd (n = 464) or
Vd (n = 465) KYPROLIS®
56 mg/m2 twice weekly.
*The primary endpoint was PFS. Patients included in these clinical trials had 1-3 prior lines of therapy.1,2,5,6
†As of the primary analysis, with a median follow-up of ~ 17 months, the primary endpoint of improved median PFS was met. Median PFS was not reached for DKd vs 15.8
months for Kd (HR = 0.63; 95% CI: 0.46–0.85; P = 0.0014, one-sided).1,4
‡As of the primary analysis, with a median follow-up of 20.7 months, the primary endpoint of improved median PFS was met. Median PFS was not reached for Isa-Kd vs
20.27 months for Kd (HR = 0.55; 95% CI: 0.37–0.82; P = 0.0032).2,3
§Analysis censoring PFS events occurring > 8 weeks from the last valid disease assessment.2
**KYPROLIS® should be discontinued after Cycle 18 when given in combination with Rd.4
CI, confidence interval; DKd, carfilzomib + daratumumab + dexamethasone; HR, hazard ratio; Isa-Kd, carfilzomib + isatuximab-irfc + dexamethasone; Kd, carfilzomib +
dexamethasone; KRd, carfilzomib + lenalidomide + dexamethasone; mPFS, median progression-free survival; NCCN, National Comprehensive Cancer Network®; PFS,
progression-free survival; Rd, lenalidomide + dexamethasone; Vd, bortezomib + dexamethasone; RRMM, relapsed or refractory multiple myeloma.
Please see accompanying full Prescribing Information.
Please see accompanying full Prescribing Information.
References: 1. Usmani SZ, Quach H, Mateos MV, et al. Carfilzomib, dexamethasone, and daratumumab versus carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma
(CANDOR): updated outcomes from a randomised, multicentre, open-label, phase 3 study. Lancet Oncol. 2022;23:65-76. 2. Martin T, Dimopoulos MA, Mikhael J, et al. Isatuximab, carfilzomib, and
dexamethasone in patients with relapsed multiple myeloma: updated results from IKEMA, a randomized Phase 3 study. Blood Cancer J. 2023;13:72. 3. SARCLISA® (isatuximab-irfc) prescribing information.
Bridgewater, NJ: sanofi-aventis U.S. 4. KYPROLIS® (carfilzomib) prescribing information, Onyx Pharmaceuticals Inc., an Amgen Inc. subsidiary. 5. Stewart AK, Rajkumar SV, Dimopoulos MA, et al. Carfilzomib,
lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015;372:142-152. 6. Dimopoulos MA, Quach H, Mateos MV, et al. Carfilzomib, dexamethasone, and daratumumab versus
carfilzomib and dexamethasone for patients with relapsed or refractory multiple myeloma (CANDOR): results from a randomised, multicentre, open-label, phase 3 study. Lancet. 2020;396:186-197.
7. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Multiple Myeloma V.5.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights
reserved. Accessed January 9, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.